The measurement of moisture content is a complex topic, but it doesn't necessarily have to be difficult. This article will elaborate on the common mistakes in trace moisture measurement, the causes of these problems, and how to avoid them.
Errors and improper operations can lead to moisture measurement results that deviate from expectations. Good sampling techniques are crucial for obtaining accurate and reliable measurement results. When designing a moisture sampling system, you should consider the following elements:
1. Material permeability
2. Adsorption and desorption
3. Sample tube length
4. Dead volume and residual moisture
5. Sample pretreatment
6. Condensation and leakage
7. Sample tube and length
8. Flow rate
Each of these items will be discussed in detail below.
Material Permeability and Humidity
All materials allow water vapor to pass through because the size of water molecules is much smaller than the structural dimensions of solids, even compared to the crystal structure of metals. The following chart shows the dew point inside pipes made of different materials when purged with very dry gas, while the outside of the pipes is in the ambient environment.
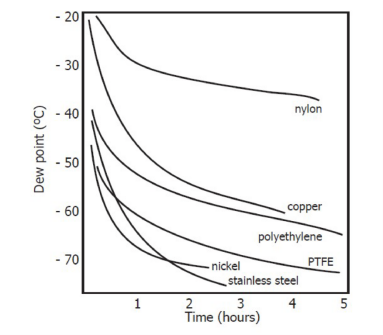
Comparison of Material Permeability
Many materials contain moisture within their structures, especially organic materials (natural or synthetic), salts (or anything containing salts), and substances with tiny pores. Therefore, it is crucial to ensure that the materials used are suitable for the specific application.
If the partial pressure of water vapor outside a compressed air pipeline is higher than that inside, water vapor from the atmosphere will naturally penetrate through the porous medium, causing water to enter the pressurized air pipeline. This phenomenon is called "transpiration."
Adsorption and Desorption
Adsorption refers to the attachment of atoms, ions, or molecules from a gas, liquid, or dissolved solid to the surface of a material, forming a thin film. The adsorption rate increases at higher pressures and lower temperatures.
Desorption refers to the release of a substance from or through the surface of a material. Under constant environmental conditions, the adsorbed substance will remain on the surface almost indefinitely. However, as the temperature rises, the likelihood of desorption also increases.
From a practical perspective, due to changes in ambient temperature, water molecules will be adsorbed and released on the inner surface of the sample tube, resulting in slight fluctuations in the measured dew point.
Sample Tube Length
The sampling point should always be as close as possible to the critical measurement point to obtain a truly representative measurement result. The length from the sensor or instrument to the sample pipeline should be as short as possible. Connection points and valves can accumulate moisture, so using the simplest sampling layout will shorten the drying time when purging the sample system with dry gas.
During the laying of longer pipelines, water will inevitably seep into any pipeline, and the effects of adsorption and desorption will become more pronounced. As is evident from the chart shown above, the materials that can effectively prevent water evaporation are stainless steel and polytetrafluoroethylene.
Dead Volume and Trapped Moisture
The dead volume in the sample pipeline (i.e., the area not in the direct flow path) can retain water molecules, which are slowly released into the passing gas; this leads to longer purging and response times, as well as readings that are more humid than expected. Hygroscopic substances in filters, valves (such as rubber components in pressure regulators), or any other components in the system may also trap moisture.
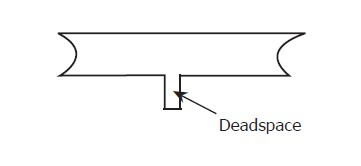
Dead volume: In experiments in fields like chemistry and biology, it refers to the volume that cannot be effectively utilized. Usually, this is because substances that cannot be mixed or react remain in this volume.
Sample Pretreatment
Sample pretreatment is often necessary to prevent sensitive measurement components from coming into contact with liquids and other contaminants. These contaminants may damage the measurement results or affect their accuracy over time, depending on the measurement technique used.
Particle filters are used to remove dirt, rust, scale, and any other solid substances from the sample flow. To prevent liquids from entering, coalescing filters should be used.
Membrane filters, although more expensive, are more effective than coalescing filters. They can prevent liquid droplets from entering and, in the event of a large amount of liquid, can completely stop the liquid from flowing to the analyzer.
Condensation and Leakage
It is crucial to keep the temperature of the sample system piping higher than the dew point temperature of the sample to prevent condensation. Any condensation will invalidate the sampling process because it changes the water vapor content in the measured gas. The liquid formed by condensation may drip or flow to other locations, where it will re - evaporate.
The integrity of all connections is also an important consideration, especially when sampling low dew points at higher pressures. If there is a small leak in a high - pressure pipeline, gas will leak out, but the vortex at the leak point and the negative vapor pressure difference can also cause water vapor to contaminate the fluid.
Flow Rate
Theoretically, the flow rate has no direct impact on the measured water content, but in practice, it may have unexpected effects on the response speed and accuracy. The optimal flow rate varies depending on the measurement technique, and this information can usually be found in the instruction manual of the instrument or sensor.
Insufficient flow rate may lead to:
- Enhanced adsorption and desorption effects as the gas passes through the sampling system.
- In complex sampling systems, allowing moisture pockets to remain undisturbed, which will then gradually mix into the sample flow.
- An increased probability of contamination caused by back - diffusion: ambient air that is more humid than the sample can flow back into the system from the exhaust port. Longer exhaust pipes (sometimes called "pigtail pipes") can also help mitigate this problem.
- A slower response of the sensor to changes in humidity content.




















 Consultation Tel.
Consultation Tel. Product Center
Product Center Solution
Solution Home
Home