Definition of dew point — What is the dew point?

Formation of dew on glass
The dew point or water dew point refers to the temperature at which air is saturated with moisture at any given pressure, causing the formation of dew, fog, or frost. It is an important indicator of humidity and plays a significant role in various applications, including oil and gas, weather forecasting, and daily life (household).
According to the National Physical Laboratory (NPL) in the UK, the dew point is defined as:
"The temperature at which dew or condensation forms when you cool a gas. When the condensate is ice, this is called the frost point."
What are the influencing factors of the dew point?
It should be noted that when the humidity reaches saturation (i.e., 100% RH) and the surface temperature is lower than the dew - point temperature, condensation occurs. The formation of condensation depends on the pressure and humidity level in the gas.
While temperature doesn't directly impact the dew point, it affects the relative humidity (rh%) and absolute humidity (Pv) of the gas.
Pressure, however, is an important variable that determines the dew point. It can influence the dew point by affecting the air's capacity to hold water vapor.
When the pressure increases, gas molecules are compressed and the gas becomes denser. In denser air or gas, water vapor molecules are more likely to collide with each other and condense into liquid water.
This means that under low vapor - pressure conditions, the gas or air can hold more water vapor before reaching 100% saturation. In contrast, a higher vapor pressure can lead to the formation of frost.
One common principle for measuring the dew point is that an increase in pressure generally raises the dew point, while a decrease in pressure lowers it.
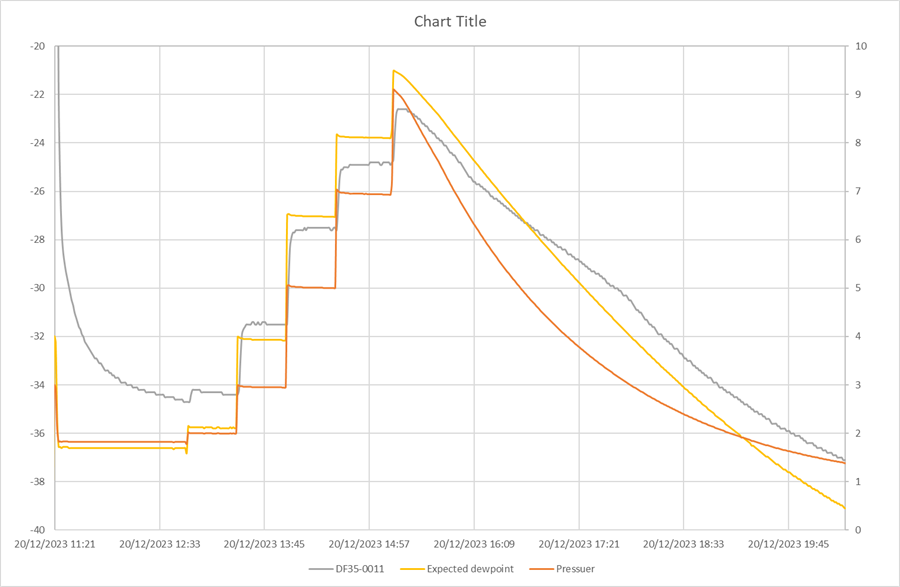
Transmitter test
The pressure was increased from 0 bar to the expected dew point, and a decrease in pressure lowered the expected dew point. This directly demonstrates the impact of pressure on the dew point when using our transmitter.
The dew point is a standard parameter in meteorology. However, in this article, we will examine why it is crucial for various industrial processes. (If you want to learn about the meteorological applications of dew point measurement, here is a brief explanation from the UK Met Office.)
Unlike other process variables such as temperature, pressure, and flow rate, moisture has two unique characteristics:
l Since humidity is directly affected by temperature and pressure, it is difficult to measure accurately.
l Moisture in the process is usually a contaminant that can seriously damage the process, equipment, and degrade product quality.
This can be prevented by using a hygrometer.
What is humidity measurement? - What is a hygrometer (dew - point meter)?
A hygrometer is a measurement of the water content in a gas. The hygrometer senses, measures, and outputs or displays the relative humidity or absolute humidity in the gas. Besides the term "hygrometer", it can also be referred to as:
l Moisture Analyzer
l Humidity sensor
l Dew-point meter
l Dew-point transmitter
l Dew-point monitor
l Moisture detector, etc.
What are the differences among humidity, dew point and relative humidity?
Humidity is simply the moisture dissolved in a gas or liquid. There is always a certain level of humidity in the air around us, but when it comes to industrial processes, the level of humidity usually needs to be controlled.
Relative humidity is a method of measuring humidity in terms of the saturation point. Just as a solid is dissolved in a liquid, a gas can only hold a certain amount of dissolved moisture before it starts to condense back into the liquid phase. The saturation point depends on the temperature and pressure of the gas. Higher temperatures and lower pressures allow more moisture to exist in the gas in the form of humidity. If the temperature decreases or the pressure increases, the saturation point changes, and the %rh also increases.
%rh It is an indicator of how close the humidity in a gas is to the saturation point.
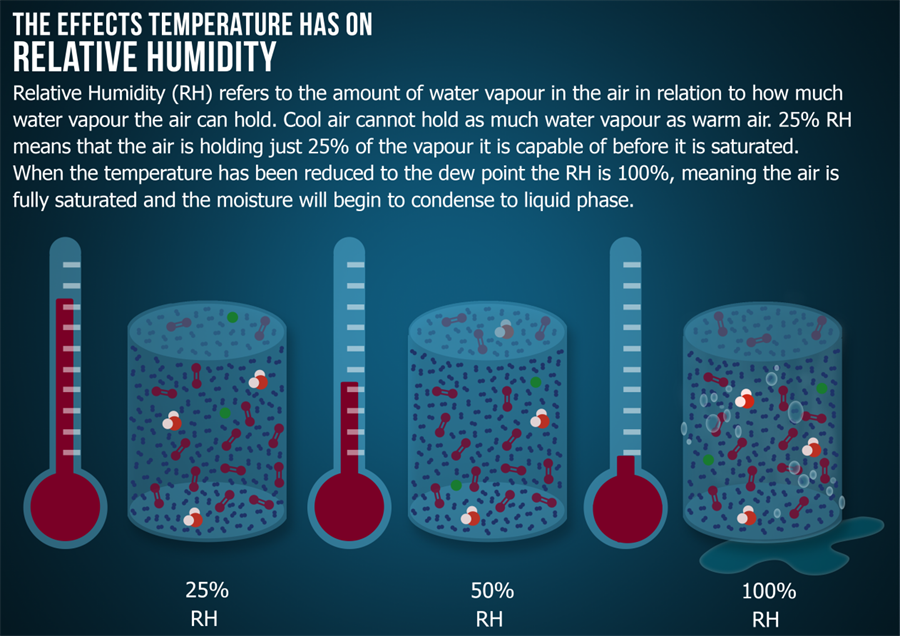
The influence of temperature on relative humidity
Moisture content Moisture content is also an important parameter for many processes. Unlike relative humidity or dew point, the moisture content does not change with pressure or temperature: it is a measurement of the actual number of water molecules in the gas.
It is expressed as parts per million by volume.(PPM V )
Dew point Dew point is another variable of humidity. Instead of looking at the water - holding capacity of a gas, the dew point measures the temperature at which liquid moisture begins to condense. Like relative humidity, the dew - point temperature depends on the pressure of the gas being measured.
It is expressed as °C dew point, °C frost point or °C td.

The impact of doubling the system pressure on the dew - point temperature
These parameters offer slightly different benefits to operators. Since the dew point is a measurement of the liquid moisture condensation temperature, it is most beneficial in applications where operators need to avoid moisture condensation—for example, in pipeline engineering, where the ambient temperature may drop below freezing. Ensuring that the dew - point temperature of the gas is lower than the lowest possible ambient temperature means that no liquid moisture freezes, and there is no risk of blockage due to icing.
The dew point is also a common parameter for monitoring the output of industrial dryers and may be specified in quality procedures.
How to calculate the dew point using the water vapor equation?
The water vapor equation you'll use depends on whether the dew point is higher or lower than 0c°C.
When the dew point exceeds 0°C, the "over - water" equation is used; similarly, if the temperature is below 0°C, water will directly condense into ice, which means the "over - ice" equation should be used. However, in some cases, a phenomenon called supercooled water may occur.
Traps in calculating the dew point - Supercooled water phenomenon and solutions
In situations where the temperature is close to but below 0°C (i.e., - 5°C), water sometimes remains in liquid form without transforming into a solid (ice). This phenomenon is called supercooled water. Calculating water vapor pressure can be problematic because most calculators for water vapor assume that a dew point below zero is "over - ice". If the condensate is liquid and the difference between the calculated water vapor pressure and the actual water vapor pressure is about 10%, then the "over - water" equation should be used.
In industries such as atmospheric metrology, since supercooling can occur in clouds and the upper atmosphere, the World Meteorological Organization (WMO) decided nearly half a century ago that RH should be calculated by calculating the saturated water vapor pressure over a water surface, regardless of the temperature. This decision was made to prevent the occasional possibility of the RH value exceeding 100% when the atmosphere is in a supercooled state.
Measurement techniques such as our chilled mirrors (Michell S8000 and Optidew) solve this problem by further reducing the temperature to ensure that the condensate is actually ice.
Dew point water vapor equation
Some of the most commonly used dew - point calculation formulas are the industrial water vapor pressure formulas by Hyland and Wexler (1983) and Sonntag (1994).
Please look at the following equations:
Hyland and Wexler (1983) below 0°C
\( \ln(e_w) = -0.58002206 \times 10^4 / T + 0.13914993 \times 10^1 - 0.48640239 \times 10^{-1} T + 0.41764768 \times 10^{-4} T^2 - 0.14452093 \times 10^{-7} T^3 + 0.65459673 \times 10^1 \times \ln(T) \)
Place:
· It is the natural logarithm of the vapor pressure over liquid water (Pa).
· \(T\)It is the temperature expressed in Kelvin (K).
Sonntag's (1994) below 0°C
\( \ln(e_w) = -\frac{6096.9385}{T} + 16.635794 - 2.711193 \times 10^{-2} T + 1.673952 \times 10-^{-5} T^2 + 2.433502 \times \lN(T) \)
Place:
· It is the natural logarithm of the vapor pressure over liquid water (hPa).
· \(T\)It is the temperature expressed in Kelvin (K).
Hyland and Wexler (1983) vapor pressure equation over the vapor pressure on ice
Hyland and Wexler (Hyland and Wexler, 1983):
og not=-0.56745359 x 104 / T
+ 0.63925247 × 101
- 0.96778430 × 10-2T
+ 0.62215701 × 10-6T2
+ 0.20747825 × 10-8T3
- 0.94840240 × 10-12T4
+ 0.41635019 × 101 logarithm(T)
with T in [K] and ei in [Pa]
Sonntag (1994) excessive vapor pressure over ice
Sunday,(1994 Year)
log not=-6024.5282/T
+ 24.721994
+ 1.0613868 × 10 -2 * T
- 1.3198825 × 10 -5 * T 2
- 0.49382577 * Log(T)
with T in [K] and ei in [hPa]
For industrial applications, the two formulas have different applicable temperature ranges. The Hyland - Wexler formula is valid in the temperature range from - 100°C to 0°C, while the Sonntag formula is applicable over a wider range from - 80°C to +50°C. This indicates that the Sonntag formula is more versatile, making it suitable for various applications.
Measuring dew point and other humidity parameters is crucial for many applications. You can use our free humidity and dew point calculator for simple humidity conversions and calculations.
Which industries measure dew point and why?
The dew point can be measured in slightly different ways in various applications, but ultimately it is used to control the humidity in a specific environment. Homeowners can measure the dew point to control the water vapor that causes mold growth or to determine the humidity level that is most comfortable for humans.
Typical dew point measurement range
According to the National Weather Service, the dew point is a good parameter to tell whether the air is dry or humid. If the dew point is below or equal to 55 degrees, the air should be dry enough to be comfortable for humans. When the dew point is between 55 and 65 degrees, the air becomes sticky, and when the dew point is greater than or equal to 65 degrees, the air tends to hold more moisture.
For industrial applications, more dry air or humidity control is required to meet standards, improve process performance, or maintain product quality.
In compressed air applications, it is very important to continuously monitor the compressed air system to comply with ISO standards that specify allowable solid particles, oil, and humidity.
The table below shows according to BCAS
|
Class
|
Vapor pressure dew point o C
|
liquid g/m 3
|
|
0
|
Specified by the equipment user, it is more stringent than Class 1.
|
|
1
|
≤ -70
|
-
|
|
2
|
≤ -40
|
-
|
|
3
|
≤ -20
|
-
|
|
4
|
≤ +3
|
-
|
|
5
|
≤ +7
|
-
|
|
6
|
≤ +10
|
-
|
|
7
|
-
|
≤ 0.5
|
|
7
|
-
|
≤ 0.5 - 5
|
|
9
|
-
|
- 10
|
|
x
|
-
|
> 10
|
Another example is natural gas and hydrocarbon applications, where trace amounts of moisture can cause pipeline rusting and hydrate formation, which may affect the process (gas flow rate) or the quality of the final product.

Rusty gas pipeline
Anti - corrosion coatings can help avoid rusting in pipelines.
However, due to other factors such as aging and wear, pipeline edges and joints, as well as environmental conditions like extreme temperatures, exposure to chemicals, or high humidity, it is not a permanent solution.
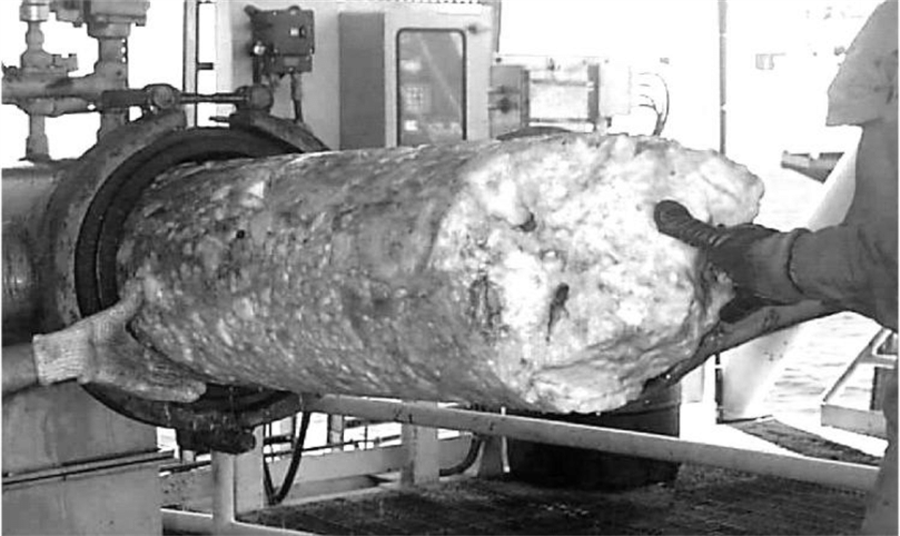
Dew point measurement to prevent hydrates from blocking pipelines
In addition to the formation of rust, when water saturation occurs in natural gas pipelines, hydrates are formed (as shown in the figure above), which may impede the flow of natural gas in the pipelines.
What is the best method for measuring dew point in industrial processes?
In industrial production processes, operators can choose from a variety of methods to measure the dew point — the final choice depends on the specific application and budget.
Dew point transmitters are small in size, cost - effective, and suitable for various safe and hazardous applications.
Portable dew - point hygrometers (dew - point meters) are best suited for testing and verification purposes. They can be used to check the readings of online moisture analyzers and also to conduct spot checks at different points in the process, such as identifying the location of leaks.
Process moisture analyzers are usually (but not always) hazardous area certified systems that include sample handling and moisture analyzers. These devices are typically designed for refineries or natural gas processing plants.
Cooled mirror hygrometers measure the actual formation of condensate on a temperature - controlled surface. They have extremely high precision and minimal drift. Although usually used as laboratory references, many models are also suitable for monitoring industrial processes, such as semiconductor manufacturing or metallurgical processes.
What are the benefits of measuring dew point in the process?
Moisture can penetrate almost any surface, invalidating test results, causing poor product quality, leading to pipeline corrosion, resulting in icing at low temperatures, causing premature wear and equipment failure, as well as reacting with other chemicals and gases.
Monitoring the dew point in process gases has the following benefits:
Process safety and equipment protection
Excessive moisture in pipelines can cause corrosion, reduce the integrity of equipment, and may lead to leaks or even explosions. Under cold conditions, ice may form and block pipelines. Controlling moisture can reduce the need for unplanned maintenance and protect employees.
Quality
Moisture has an adverse effect on many final products. In metallurgy, the humidity in the furnace needs to be carefully controlled to prevent product weakening. In pharmaceutical production, powders must be kept dry to avoid caking. Refineries need to maintain low humidity to avoid unwanted chemical reactions.
Energy conservation
The highest cost for many processes is the heat required by the dryer. Monitoring the dryer's output can ensure that the cycle stops immediately after the drying cycle is completed.
Meeting rules
Many global industrial institutions agree on international quality standards, which usually include moisture or dew - point levels. An example of this is the EASEE natural gas quality standard.
























 Consultation Tel.
Consultation Tel. Product Center
Product Center Solution
Solution Home
Home