Humidity is defined as a certain measure of the water vapor content in air (or other gases). "Humidity" is a general term used to quantify the amount of water vapor in a gas.
The term "humidity" is often used interchangeably with "relative humidity", but in the context of precise measurements, there are significant differences between the two terms.
This chapter explains why the difference between these two terms is important for those involved in the business of measuring water vapor in sensitive environments, and covers the terms and definitions used to quantify the amount of water vapor in a gas.
Water vapor plays a crucial role in maintaining the quality and efficacy of products that enhance our daily lives – products that we consumers trust are manufactured to specification. For manufacturers in various industries, it is essential to understand how accurate humidity measurement works and the role of measuring instruments.
The Importance of Accuracy
Humidity is measured using a hygrometer, a tool that utilizes various materials and measurement methods to assess the water vapor levels in a room or space. While no scientific measurement is absolutely accurate, obtaining humidity measurements as accurately as possible is crucial for various industries. Since water vapor above a certain level can cause condensation, which can ultimately lead to corrosion or mold, highly accurate humidity measurement is essential for preventing the deterioration of a wide range of items, including wooden building materials, food, pharmaceuticals, fuels, paper, electronic components, and many other materials. Humidity measurement helps maintain the optimal environmental conditions for products and prevents costly damage to valuable items.
Technical Information
In this section, we will reveal the basic physical laws governing relative humidity. In the temperature range from -50°C to 150°C and at pressures not exceeding 1000 kPa, water vapor actually behaves like an ideal gas. We will illustrate with examples the effects of temperature and pressure on relative humidity, as well as how to convert relative humidity to dew point and absolute humidity.
First, let's introduce the basics by reviewing the general properties of water vapor in moist air.
Properties of Water Vapor in Moist Air
Evaporation
When water molecules leave a surface and form a gas, they have evaporated. By absorbing or releasing kinetic energy, water molecules change from the liquid state to the gaseous state. The liquid water that turns into water vapor takes away a portion of heat through the process of evaporative cooling.
Definition:
Evaporative cooling is the reduction in air temperature caused by the evaporation of a liquid, removing heat from the surface where evaporation occurs. The energy removed during the evaporative cooling process is called "latent heat".
Evaporative cooling is constrained by atmospheric conditions. When the air is very hot and dry, the evaporation process consumes more heat, making the cooling effect more significant than when evaporative cooling occurs in hot and humid air.
Condensation
The process by which water vapor turns into a liquid is called condensation. Water vapor will only condense on a surface when the surface temperature is below the dew point temperature or when the water vapor equilibrium in the air is exceeded. When water vapor freezes on a surface, a net warming occurs. The water molecules release heat, which in turn causes a slight increase in the atmospheric temperature.
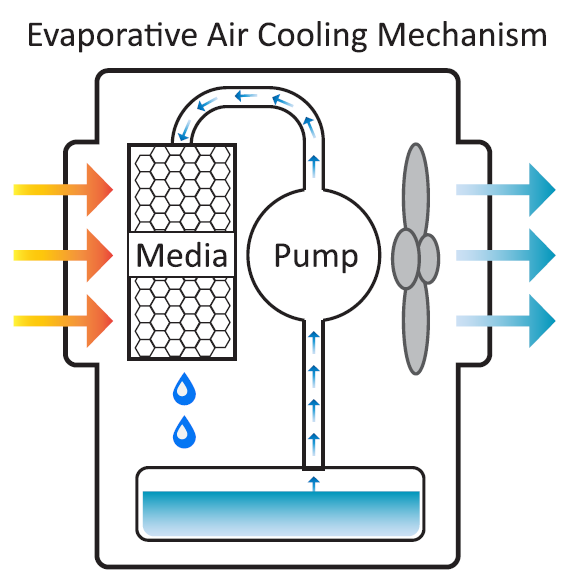
"Swamp coolers" regulate air in hot weather through water evaporation. They work well in dry climates like Denver or Phoenix, but not as effectively in Houston or Boston, where the air is more humid.
Chemical Reactions
Many chemical reactions produce water as a product. If the reaction occurs at a temperature higher than the dew point of the surrounding air, water forms vapor and increases the amount of water vapor in the gas. If the reaction takes place at an air temperature below the dew point, condensation occurs and water vapor leaves the gas.
Other chemical reactions occur in the presence of water vapor, leading to the formation of new chemical substances, such as rust on iron or steel.



















 Consultation Tel.
Consultation Tel. Product Center
Product Center Solution
Solution Home
Home