Water vapor is one of the several gases that make up air. For example, if the total pressure of a system (such as air at sea level) is 101.3 kPa (or 29.9 inches of mercury), and the air consists of nitrogen, oxygen, water vapor, and other trace gases, each of these gases contributes to the total pressure of 101.3 kPa. The portion contributed by water vapor is called the partial pressure of water vapor. The partial pressure of water vapor is a key metric in the formulas that define all other humidity parameters.
The Impact of Pressure Changes
Dalton's Law:
According to Dalton's Law, the total pressure of a gas mixture is equal to the sum of the partial pressures of its components. This means that the partial pressure of a component is equal to the product of the total pressure and the mole fraction of the component.
Therefore, a change in the total pressure of a gas mixture without changing its composition will cause a change in the partial pressure of each component. For example, doubling the total pressure of a gas mixture will double the partial pressure of each component. As the total pressure increases, the partial pressure of water vapor will increase accordingly. This is an important fact to understand, as you will see when defining relative humidity and dew point temperature. An increase in the pressure of a closed system will increase the relative humidity and raise the dew point temperature until saturation is reached.
Vapor Pressure above a Liquid
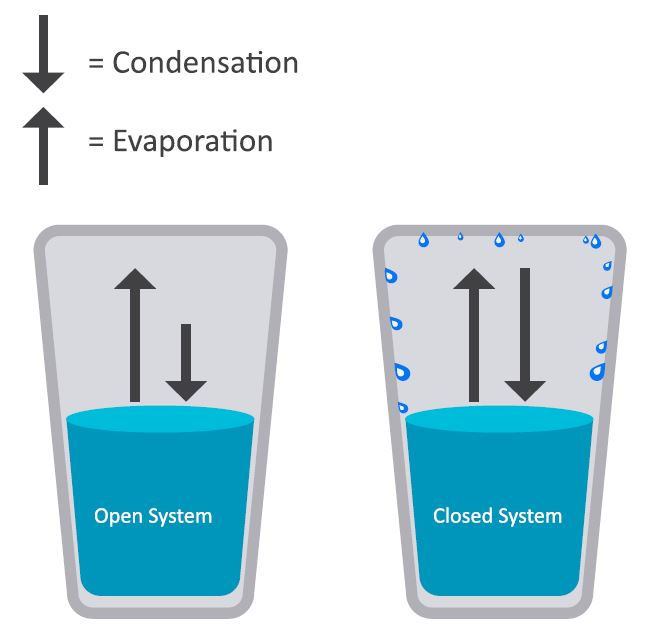
Since the molecules in a liquid are closer together than those in a gas, the intermolecular forces are stronger than in a gas. For a liquid to vaporize, the intermolecular forces must be overcome by the kinetic energy of the molecules. If a liquid is placed in a closed container, the particles that enter the gas phase cannot escape. In their random motion, the particles strike the liquid and are recaptured by the intermolecular forces. Thus, two processes occur simultaneously: evaporation and condensation. The rate of evaporation increases as the temperature rises. This is because an increase in temperature corresponds to an increase in the kinetic energy of the molecules. At the same time, as the number of particles in the gas phase increases, the rate of condensation increases: more molecules strike the liquid surface.
When these two processes are equal, the number of particles and the pressure in the gas phase stabilize.
The value of the equilibrium vapor pressure depends on the attractive forces between the liquid particles and the temperature of the liquid. The vapor pressure above a liquid increases as the temperature rises.
Saturated Water Vapor Pressure
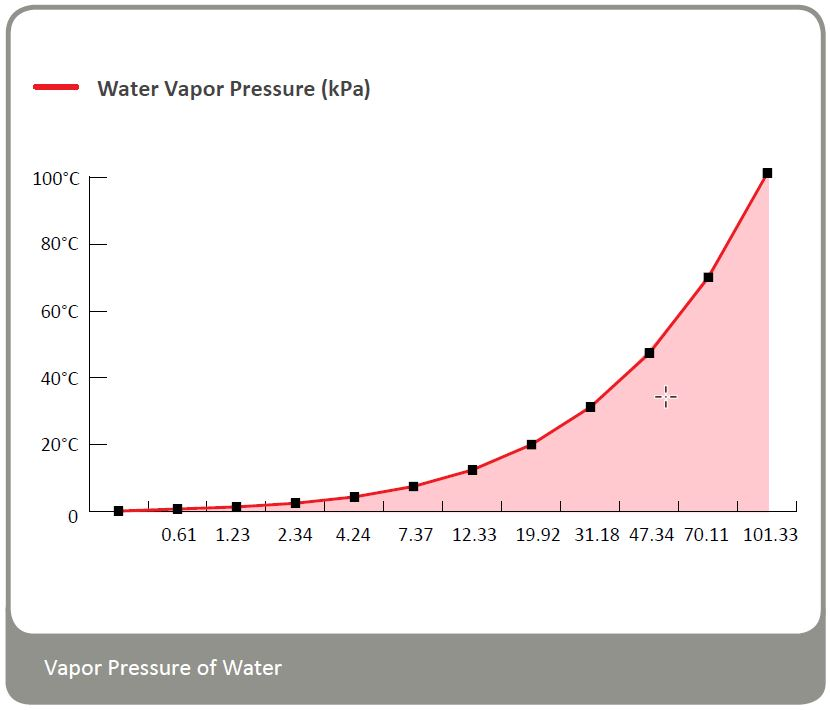
The vapor pressure of water strongly saturates as the temperature rises.
Vapor Pressure over Ice
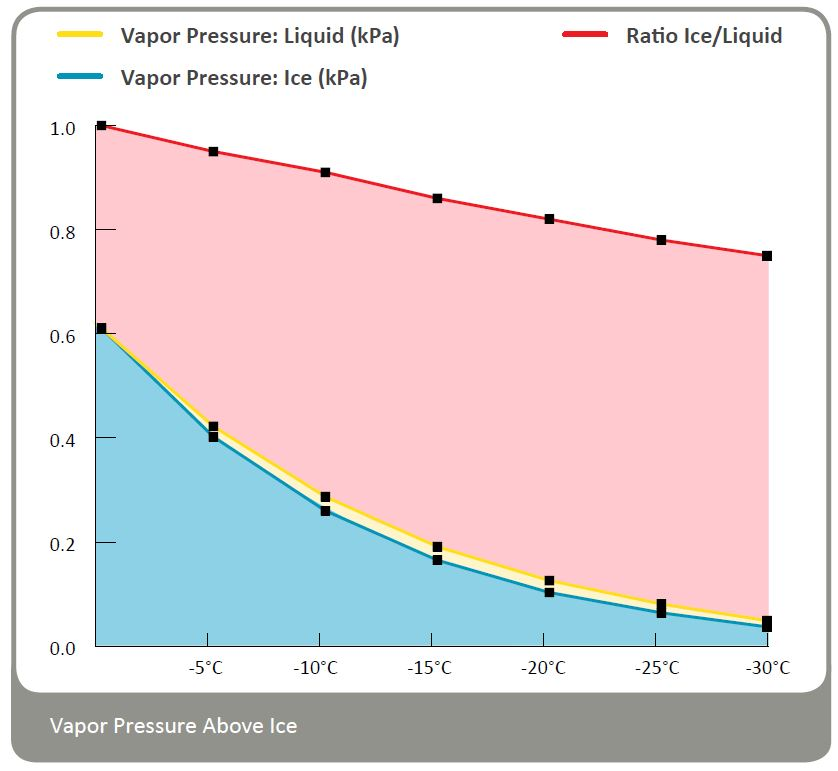
When water freezes, the molecules assume a structure that allows for the maximum number of hydrogen - bonding interactions between them. Due to the presence of large hexagonal pores in this structure, ice is more open and less dense than liquid water. Since the hydrogen bonds in ice are stronger than those in liquid water, the intermolecular attractive forces are the strongest in ice. That's why the vapor pressure over ice is lower than that over liquid water.





















 Consultation Tel.
Consultation Tel. Product Center
Product Center Solution
Solution Home
Home